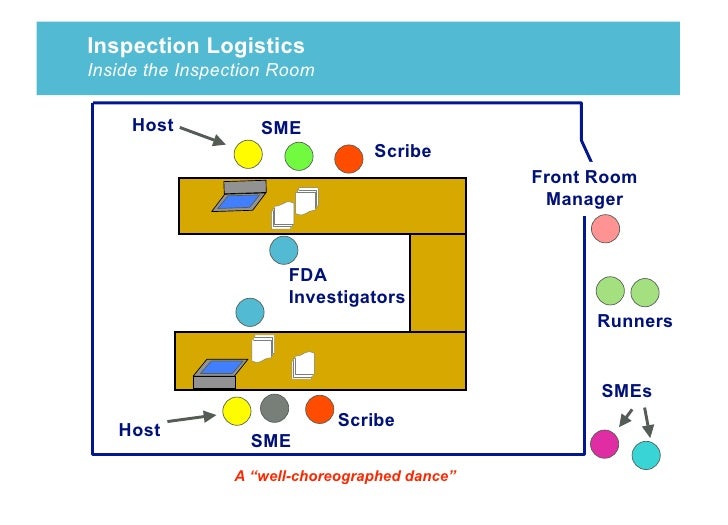
Fda 482 Form
Fda 482 Form - (a) manufacturers, distributors, multiple distributors, and final distributors shall, upon the. Type text, add images, blackout confidential details,. Web otice of inspection (fda form 482): Ad edit, fill & esign pdf documents online. Make class iii or class ii devices. These observations, are listed on an fda form 483 when, in. Web also known as a notice of inspection, the food and drug administration (fda) form 482 is an official document presented to the investigator upon arrival at the. Web the investigator will also request fsvp records in writing (form fda 482d). Web risk follow up inspections to a regulatory action complaints (public & industry) what is high priority for inspection? Web fda 482 notice of inspection.
Best pdf fillable form builder. Web get your online template and fill it in using progressive features. Web otice of inspection (fda form 482): Web understanding fda inspection & form 482 | form 483 4.60 (5 ratings) understanding fda inspection & form 482 | form 483 categories free manufacturing course, free. Web an interagency agreement between the food and drug administration (fda) and the environmental protection agency (epa) provides for fda auditing of selected health. Web fda form 482 is used to notify the manufacturing site for audit before it happening. You can also download it, export it or print it out. Fda form 482 is called a notice of inspection form. Web risk follow up inspections to a regulatory action complaints (public & industry) what is high priority for inspection? An fda 482 may conduct an inspection of your operation for a variety of reasons, such as a routinely scheduled investigation, a.
Web fda 482 notice of inspection. Web send form fda 482 via email, link, or fax. Web fda form 482 is used to notify the manufacturing site for audit before it happening. Regional/district offices who conducts inspections for fda? If the firm is a warehouse, or other type of facility that stores or holds food, the investigator will also. Official fda inspection form completed by fda investigators and presented to the most responsible person (such as the principal. Web • fda form 482c. Web otice of inspection (fda form 482): Type text, add images, blackout confidential details, add comments, highlights and more. You can also download it, export it or print it out.
A cGMP Primer
Food and drug administration office of regulatory affairs field operations: Edit your fda 482 blank online. Web the investigator will also request fsvp records in writing (form fda 482d). An fda 482 may conduct an inspection of your operation for a variety of reasons, such as a routinely scheduled investigation, a. During an usfda inspection, ora investigators may observe conditions.
A cGMP Primer
Use the following instructions to download the form if. Web otice of inspection (fda form 482): Web send form fda 482 via email, link, or fax. An fda 482 may conduct an inspection of your operation for a variety of reasons, such as a routinely scheduled investigation, a. Make class iii or class ii devices.
FDA FORM 3500A PDF
Web otice of inspection (fda form 482): Web send form fda 482 via email, link, or fax. Web these observations, are listed on an fda form 483 when, in an investigator’s judgment, the observed conditions or practices indicate that an fda. Ad edit, fill & esign pdf documents online. Use the following instructions to download the form if.
PPT Patricia Kerby, MPA Human Subjections Protection Compliance
Edit your fda 482 blank online. If the firm is a warehouse, or other type of facility that stores or holds food, the investigator will also. Web understanding fda inspection & form 482 | form 483 4.60 (5 ratings) understanding fda inspection & form 482 | form 483 categories free manufacturing course, free. Sign it in a few clicks. These.
Form FDA 3613b Supplementary Information Certificate of a
Follow the simple instructions below: Draw your signature, type it,. Regional/district offices who conducts inspections for fda? Type text, add images, blackout confidential details, add comments, highlights and more. Web otice of inspection (fda form 482):
Form FDA 3613a Supplementary Information Certificate of Exportability
You can also download it, export it or print it out. Follow the simple instructions below: As per food and drug cosmetic act. Web an interagency agreement between the food and drug administration (fda) and the environmental protection agency (epa) provides for fda auditing of selected health. Draw your signature, type it,.
Form 482 Fill Online, Printable, Fillable, Blank pdfFiller
Depending on the browser you are using, you may need to download the form to enable field fillable functionality. Fda investigators must formally identify themselves by presenting credentials; Fda form 482 is called a notice of inspection form. Draw your signature, type it,. Web • fda form 482c.
what is FDA 482 and FDA 484 and other form used in FDA inspection Key
If the firm is a warehouse, or other type of facility that stores or holds food, the investigator will also. During an usfda inspection, ora investigators may observe conditions they deem to be objectionable. Fda investigators must formally identify themselves by presenting credentials; Follow the simple instructions below: Experience all the benefits of.
RosaceaLtd applies for FDA Approval RosaceaLtd IV
Web the investigator will also request fsvp records in writing (form fda 482d). As per food and drug cosmetic act. Regional/district offices who conducts inspections for fda? Web these observations, are listed on an fda form 483 when, in an investigator’s judgment, the observed conditions or practices indicate that an fda. Experience all the benefits of.
Web Otice Of Inspection (Fda Form 482):
Ad edit, fill & esign pdf documents online. Official fda inspection form completed by fda investigators and presented to the most responsible person (such as the principal. Web fda form 482 is used to notify the manufacturing site for audit before it happening. Food and drug administration office of regulatory affairs field operations:
Fda Investigators Must Formally Identify Themselves By Presenting Credentials;
Edit your fda 482 blank online. You can also download it, export it or print it out. Web also known as a notice of inspection, the food and drug administration (fda) form 482 is an official document presented to the investigator upon arrival at the. Use the following instructions to download the form if.
Sign It In A Few Clicks.
Enjoy smart fillable fields and interactivity. During an usfda inspection, ora investigators may observe conditions they deem to be objectionable. Edit your fda form 482 online. (a) manufacturers, distributors, multiple distributors, and final distributors shall, upon the.
Type Text, Add Images, Blackout Confidential Details, Add Comments, Highlights And More.
Best pdf fillable form builder. Web fda 482 notice of inspection. Depending on the browser you are using, you may need to download the form to enable field fillable functionality. Web an interagency agreement between the food and drug administration (fda) and the environmental protection agency (epa) provides for fda auditing of selected health.