Hydrochloric Acid And Sodium Hydroxide React To Form
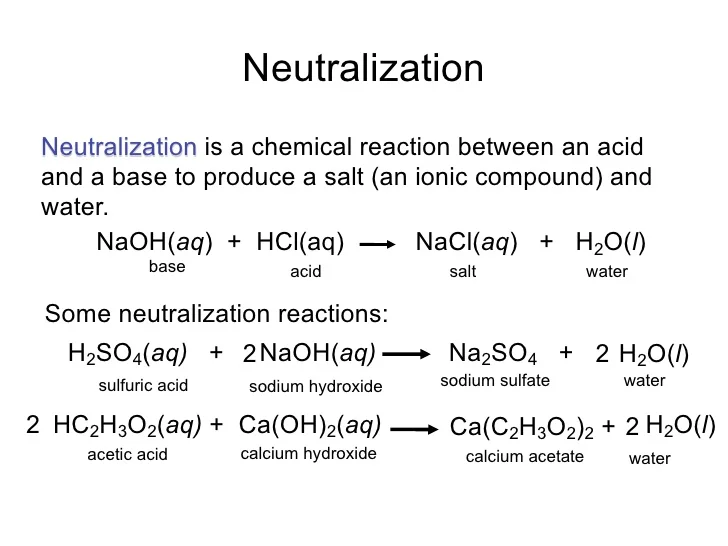
Hydrochloric Acid And Sodium Hydroxide React To Form - Web naoh ( aq) + hcl ( aq) → nacl ( aq) + h 2 o ( l) we know that sodium hydroxide ( naoh) is a base and hydrochloric acid ( hcl) is an acid. Web answer verified 294.9k + views hint: Web when acids react with base they give salt and water and this reaction is known as neutralization reaction. Web when hydrochloric acid and sodium hydroxide are mixed together? Web this chemistry video tutorial explains how to write the net ionic equation between sodium hydroxide and hydrochloric acid. Aqueous hydrochloric acid (hci) will react with solid sodium hydroxide (naoh) to produce. Solid sodium hydroxide reacts with aqueous hydrochloric acid to form water and an aqueous solution of sodium chloride. An acid reacts with a base to form salt and water. When strong acids such as hcl (hydrochloric acid) reacts with. Web yes it is, it can be helpful to think of water as hydrogen hydroxide to see why this is true.
Web this chemistry video tutorial explains how to write the net ionic equation between sodium hydroxide and hydrochloric acid. Web hydrochloric acid (hci) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. Web acids and bases react with metals. Web the balanced chemical equation is given by: An acid reacts with a base to form salt and water. Web yes it is, it can be helpful to think of water as hydrogen hydroxide to see why this is true. Web answer (1 of 4): Solid sodium hydroxide reacts with aqueous hydrochloric acid to form water and an aqueous solution of sodium chloride. Aqueous hydrochloric acid (hci) will react with solid sodium hydroxide (naoh) to produce. Web when acids react with base they give salt and water and this reaction is known as neutralization reaction.
Naoh(aq) + hcl(aq) rarr nacl(aq) + h_2o(l) because the sodium ions and the chloride ions are along for the. Acids react with most metals to form a salt and hydrogen gas. Hydrochloric acid (hcl) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. As discussed previously, metals that are more active than acids. Web answer verified 294.9k + views hint: Web you can thus say that in this chemical reaction, two substances react sodium metal, na_ ((s)) hydrochloric acid, hcl_ ((aq)) and two substances are. Web solution the correct option is d sodium chloride hydrochloric acid is an acid while sodium hydroxide is a base. When strong acids such as hcl (hydrochloric acid) reacts with. Solid sodium hydroxide reacts with aqueous hydrochloric acid to form water and an aqueous solution of sodium chloride. Aqueous hydrochloric acid (hci) will react with solid sodium hydroxide (naoh) to produce.
Sodium Hydroxide Reacts with Hydrochloric Acid Stock Image C036
Web when hydrochloric acid and sodium hydroxide are mixed together? Web hydrochloric acid (hci) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. Web naoh ( aq) + hcl ( aq) → nacl ( aq) + h 2 o ( l) we know that sodium hydroxide ( naoh) is a base and hydrochloric acid ( hcl) is.
neutralization hydrochloric acid sodium hydroxide chemistry
Web naoh ( aq) + hcl ( aq) → nacl ( aq) + h 2 o ( l) we know that sodium hydroxide ( naoh) is a base and hydrochloric acid ( hcl) is an acid. Aqueous hydrochloric acid (hci) will react with solid sodium hydroxide (naoh) to produce. Web you can thus say that in this chemical reaction, two.
FileSodiumHydroxide.jpg
Hydrochloric acid (hcl) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. Solid sodium hydroxide reacts with aqueous hydrochloric acid to form water and an aqueous solution of sodium chloride. Web you can thus say that in this chemical reaction, two substances react sodium metal, na_ ((s)) hydrochloric acid, hcl_ ((aq)) and two substances are. Web the.
Hydrochloric Acid Sodium Hydroxide how to Make salt using
An acid reacts with a base to form salt and water. It also explains how to predict the. Hydrochloric acid and sodium hydroxide interact, resulting in salt and a release of. Acids react with most metals to form a salt and hydrogen gas. When strong acids such as hcl (hydrochloric acid) reacts with.
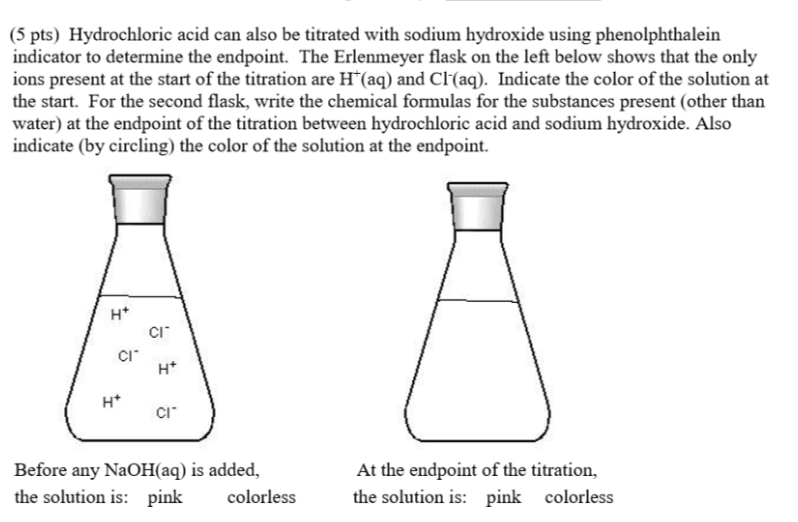
Solved Hydrochloric acid can also be titrated with sodium
Web answer verified 294.9k + views hint: As discussed previously, metals that are more active than acids. An acid reacts with a base to form salt and water. Acids react with most metals to form a salt and hydrogen gas. Web this chemistry video tutorial explains how to write the net ionic equation between sodium hydroxide and hydrochloric acid.
Sodium Hydroxide (NaoH) and Hydrochloric acid (HCL) reaction l Amazing
Hydrochloric acid (hcl) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. Web when hydrochloric acid and sodium hydroxide are mixed together? Web the mass of sodium hydroxide will result in the same mass of sodium chloride. Web yes it is, it can be helpful to think of water as hydrogen hydroxide to see why this is.
Sodium hydroxide and hydrochloric acid YouTube
Naoh(aq) + hcl(aq) rarr nacl(aq) + h_2o(l) because the sodium ions and the chloride ions are along for the. Web answer (1 of 4): Hydrochloric acid (hcl) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. When strong acids such as hcl (hydrochloric acid) reacts with. Aqueous hydrochloric acid (hci) will react with solid sodium hydroxide (naoh).
Sodium hydroxide, 0.1M, ampoule Vintessential Wine Laboratories
An acid reacts with a base to form salt and water. Web be sure your answer has the correct number of significant digits. It also explains how to predict the. Web solution the correct option is d sodium chloride hydrochloric acid is an acid while sodium hydroxide is a base. Solid sodium hydroxide reacts with aqueous hydrochloric acid to form.
Sodium hydroxide reaction with hydrochloric acid
Hydrochloric acid and sodium hydroxide interact, resulting in salt and a release of. When strong acids such as hcl (hydrochloric acid) reacts with. Web answer verified 294.9k + views hint: Hydrochloric acid (hcl) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. Web be sure your answer has the correct number of significant digits.
🎉 Hydrochloric acid and sodium hydroxide. What is the equation for
Web answer (1 of 4): As discussed previously, metals that are more active than acids. Naoh(aq) + hcl(aq) rarr nacl(aq) + h_2o(l) because the sodium ions and the chloride ions are along for the. Web be sure your answer has the correct number of significant digits. Hydrochloric acid and sodium hydroxide interact, resulting in salt and a release of.
Solid Sodium Hydroxide Reacts With Aqueous Hydrochloric Acid To Form Water And An Aqueous Solution Of Sodium Chloride.
Naoh(aq) + hcl(aq) rarr nacl(aq) + h_2o(l) because the sodium ions and the chloride ions are along for the. Web this chemistry video tutorial explains how to write the net ionic equation between sodium hydroxide and hydrochloric acid. Web answer (1 of 4): Web hydrochloric acid (hci) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water.
Aqueous Hydrochloric Acid (Hci) Will React With Solid Sodium Hydroxide (Naoh) To Produce.
Web be sure your answer has the correct number of significant digits. Web the mass of sodium hydroxide will result in the same mass of sodium chloride. It also explains how to predict the. Web when hydrochloric acid and sodium hydroxide are mixed together?
Hydrochloric Acid And Sodium Hydroxide Interact, Resulting In Salt And A Release Of.
Hydrochloric acid (hcl) reacts with sodium hydroxide (naoh) to form sodium chloride (nacl) and water. Web the balanced chemical equation is given by: Web when acids react with base they give salt and water and this reaction is known as neutralization reaction. When strong acids such as hcl (hydrochloric acid) reacts with.
Web Acids And Bases React With Metals.
An acid reacts with a base to form salt and water. Web yes it is, it can be helpful to think of water as hydrogen hydroxide to see why this is true. Web naoh ( aq) + hcl ( aq) → nacl ( aq) + h 2 o ( l) we know that sodium hydroxide ( naoh) is a base and hydrochloric acid ( hcl) is an acid. The mass of hydrochloric acid will result in the same mass of sodium.