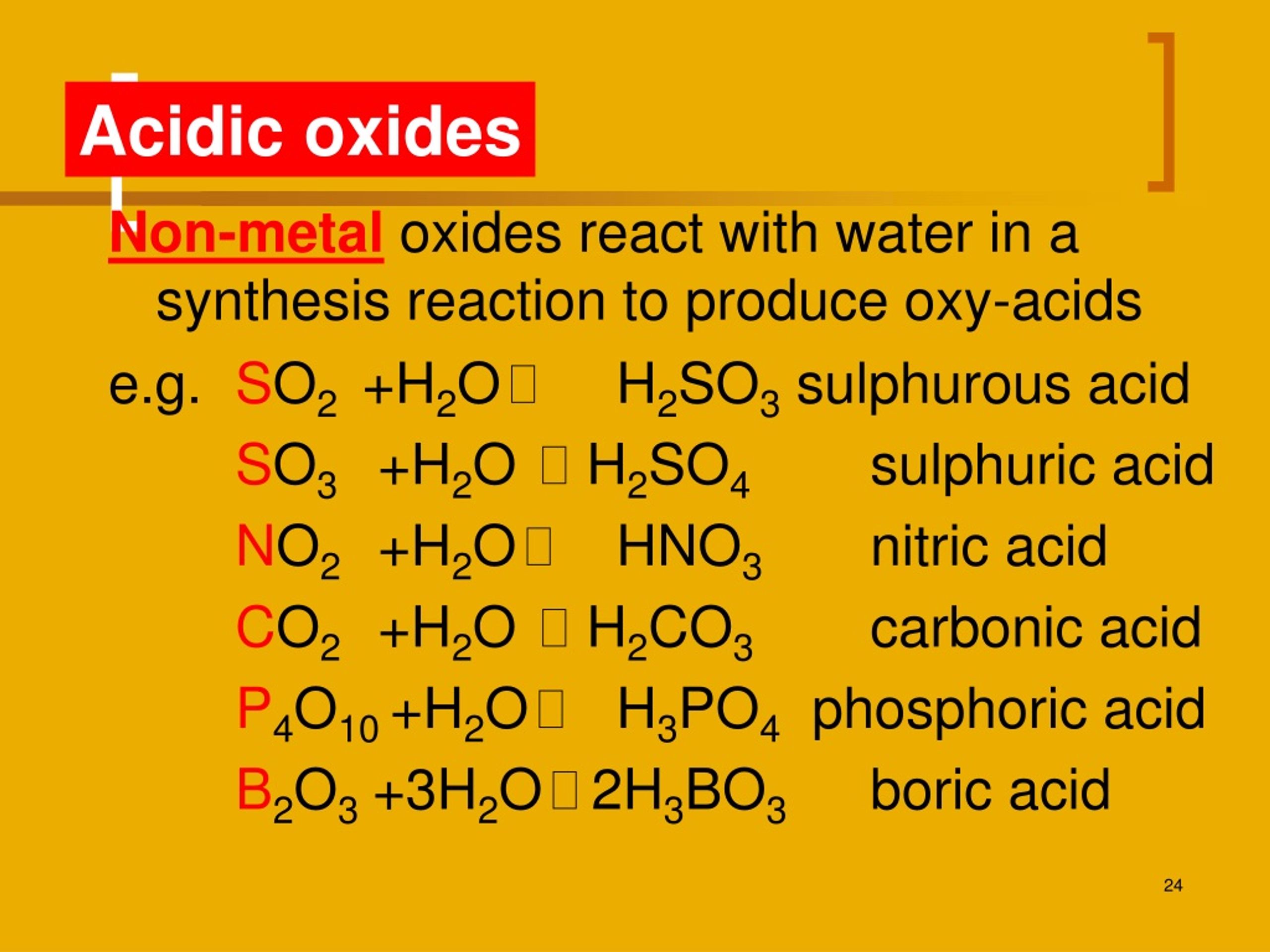
Metal Oxides React With Water To Form
Metal Oxides React With Water To Form - Here metal displaces hydrogen in acid and forms a salt. Web metal oxides and metal hydroxides are basic in nature. If soluble in water, they react with water to produce hydroxides (alkalies) e.g., cao+ h 2o → ca(oh) 2 mgo+ h 2o →. Now here is my question: Web easy solution verified by toppr correct option is a) metals react with water to form metal oxides or metal hydroxide (froming an alkaline solution) and hydrogen gas. (1) write an equation to show how barium oxide reacts with water to form. Web reaction with water: Web explanations (including important chemical equations): Examples of such reactions are: So they react with acid to form salt and water.
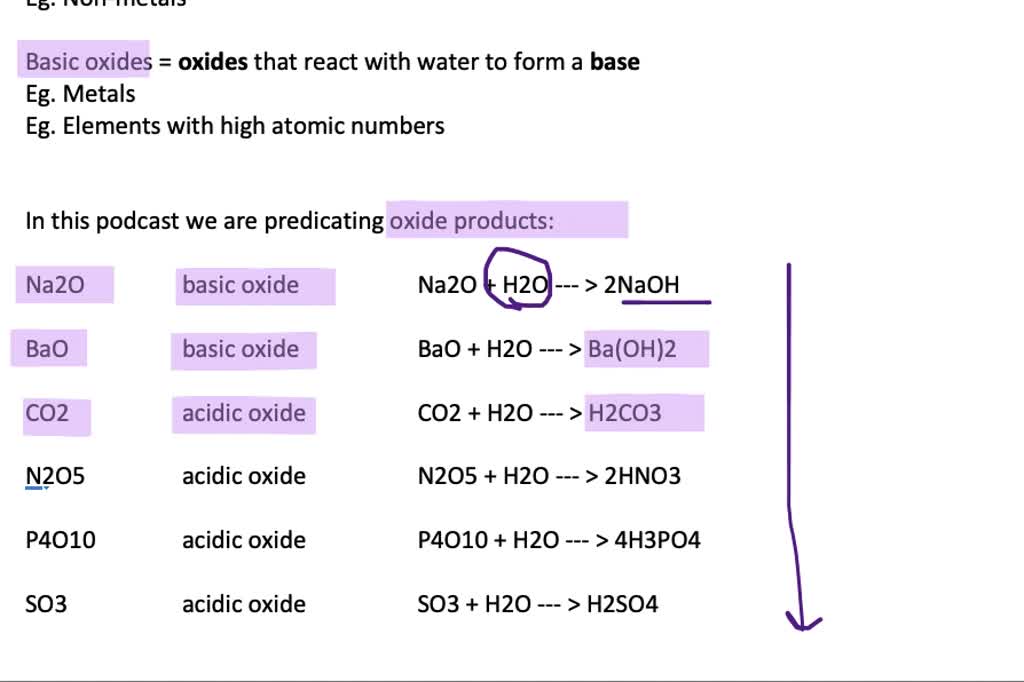
The reactivity of the metal determines which reactions the metal participates in. Web the oxides of the metals of groups 1 and 2 and of thallium(i) oxide react with water and form hydroxides. Most of the metal oxides are insoluble in water, but oxides of alkali metals and alkaline earth metals are soluble in water. Web some metal oxides are amphoteric because they can react with both acids and bases to produce salt and water. While most metal oxides are insoluble in water, alkali metal and alkaline earth metal oxides are water soluble. A concentrated solution of sodium oxide in water. Watch this video to see a. Web science chemistry chemistry questions and answers metal oxides can react with water to form bases. Web solution the correct option is b bases metal oxides react with water to form metal hydroxides which are basic in nature. Examples of such reactions are:
Web the reaction of metal oxides with water. (1) write an equation to show how potassium oxide reacts with water to form a base. Web explanations (including important chemical equations): Web metal oxides’ interaction with water. Metal oxides (to the left of the periodic table): (1) write an equation to show how barium oxide reacts with water to form. Web reaction with water: Web as far as i know, only metal oxides from group 1 and 2 react in water, making hydroxides. Metal oxides can react with water to form bases. Web metal oxides and metal hydroxides are basic in nature.
How acids react with metallic oxides CBSE Class 10 Chemistry Notes
Now here is my question: For example, magnesium oxide reacts with water. This is because of the unique nature and bonding in the oxide. The reactivity of the metal determines which reactions the metal participates in. Here metal displaces hydrogen in acid and forms a salt.
PPT Working With Chemical Reactions PowerPoint Presentation ID3661856
Web explanations (including important chemical equations): Metal oxides (to the left of the periodic table): Web the oxides of the metals of groups 1 and 2 and of thallium(i) oxide react with water and form hydroxides. While most metal oxides are insoluble in water, alkali metal and alkaline earth metal oxides are water soluble. Metal oxides can react with water.
[MCQ] Which of the statements is not correct? All metal oxides react
(1) write an equation to show how potassium oxide reacts with water to form a base. Watch this video to see a. Web metals generally form basic oxides. Sodium oxide, na 2 o, and magnesium oxide, mgo, are made up of ions. Web basic oxides are the oxides of metals.
Metal oxides can react with water to form bases.
Metal oxides that dissolve in water react with water to form basic solutions. Web as far as i know, only metal oxides from group 1 and 2 react in water, making hydroxides. For example, magnesium oxide reacts with water. The reactivity of the metal determines which reactions the metal participates in. Here metal displaces hydrogen in acid and forms a.
Difference Between Metal Oxides and Non Metal Oxides Definition
Web solution the correct option is b bases metal oxides react with water to form metal hydroxides which are basic in nature. This is because of the unique nature and bonding in the oxide. While most metal oxides are insoluble in water, alkali metal and alkaline earth metal oxides are water soluble. Sodium oxide, na 2 o, and magnesium oxide,.
[MCQ] Which of the statements is not correct? All metal oxides react
Web easy solution verified by toppr correct option is a) metals react with water to form metal oxides or metal hydroxide (froming an alkaline solution) and hydrogen gas. If soluble in water, they react with water to produce hydroxides (alkalies) e.g., cao+ h 2o → ca(oh) 2 mgo+ h 2o →. Metal oxides can react with water to form bases..
PPT Chemical Equations and Reactions PowerPoint Presentation, free
Sodium oxide, na 2 o, and magnesium oxide, mgo, are made up of ions. Here metal displaces hydrogen in acid and forms a salt. Examples include the reaction of cobalt(ii) oxide. If i add hcl to the hydroxide, can i be sure. Na 2 o ( s ) + h 2 o ( l ) naoh ( a q ).
Metals and non metals
For example, magnesium oxide reacts with water. Watch this video to see a. Nonmetals generally form acidic oxides. Web solution the correct option is b bases metal oxides react with water to form metal hydroxides which are basic in nature. Web reactions of metals metals can react with water, acid and oxygen.
Nature of metal oxides Metals and Non metals Chemistry Khan
Web metal oxides and metal hydroxides are basic in nature. Web as far as i know, only metal oxides from group 1 and 2 react in water, making hydroxides. Web the oxides of the metals of groups 1 and 2 and of thallium(i) oxide react with water and form hydroxides. Web behaviour of the period 3 oxides with water. Web.
Oxides RASILL
Watch this video to see a. While most metal oxides are insoluble in water, alkali metal and alkaline earth metal oxides are water soluble. Web reaction with water: If soluble in water, they react with water to produce hydroxides (alkalies) e.g., cao+ h 2o → ca(oh) 2 mgo+ h 2o →. Examples of such reactions are:
Most Of The Metal Oxides Are Insoluble In Water, But Oxides Of Alkali Metals And Alkaline Earth Metals Are Soluble In Water.
Here metal displaces hydrogen in acid and forms a salt. Web metals generally form basic oxides. Na 2 o ( s ) + h 2 o ( l ) naoh ( a q ). Web science chemistry chemistry questions and answers metal oxides can react with water to form bases.
Nonmetals Generally Form Acidic Oxides.
So they react with acid to form salt and water. Examples include the reaction of cobalt(ii) oxide. For example, magnesium oxide reacts with water. Watch this video to see a.
Sodium Oxide Reacts Exothermically With Cold Water To Produce Sodium Hydroxide Solution.
Web metal oxides and metal hydroxides are basic in nature. Metal oxides can react with water to form bases. Sodium oxide, na 2 o, and magnesium oxide, mgo, are made up of ions. Web basic oxides are the oxides of metals.
While Most Metal Oxides Are Insoluble In Water, Alkali Metal And Alkaline Earth Metal Oxides Are Water Soluble.
If i add hcl to the hydroxide, can i be sure. Web some metal oxides are amphoteric because they can react with both acids and bases to produce salt and water. This is because of the unique nature and bonding in the oxide. Web the reaction of metal oxides with water.


![[MCQ] Which of the statements is not correct? All metal oxides react](https://d1avenlh0i1xmr.cloudfront.net/large/0146fde4-9748-4104-875a-ccbefb146ee1/reaction-of-metal-carbonate-with-acid---teachoo-01.jpg)

![[MCQ] Which of the statements is not correct? All metal oxides react](https://d1avenlh0i1xmr.cloudfront.net/1942b2cf-3ca7-4b5e-9607-79be588ba41c/reaction-of-non-metals-oxides-with-water---teachoo.jpg)