What Type Of Elements Form Ionic Bonds With Metals
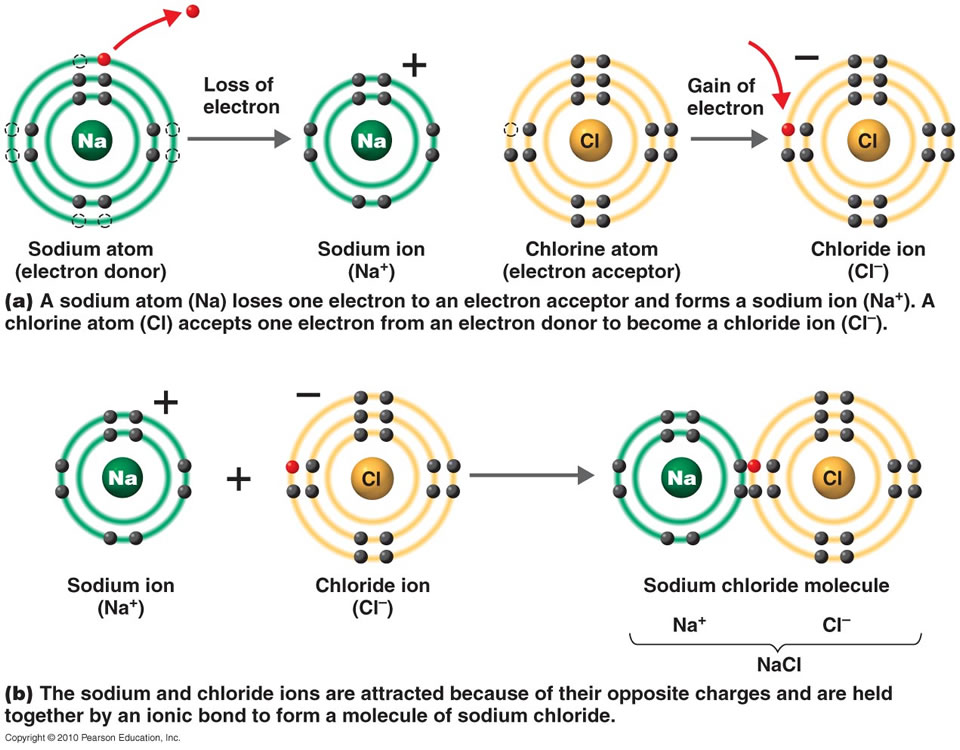
What Type Of Elements Form Ionic Bonds With Metals - This exchange results in a more stable, noble gas. The chemical bond that is formed between 2 2 atoms through the transfer of one or more electrons from the electropositive or metallic element. Web ionic bonds are one of the two main types of chemical bonds. Metals on the left and in the center of the periodic table form ionic bonds with nonmetals on the right of the. They form as a result of electrostatic attraction between oppositely charged ions and usually occur. Web the three ions would adhere (bond) to each other by the positive/negative attraction between the ions. For example, sodium (na), a metal, and chloride (cl), a nonmetal, form an ionic bond to make nacl. Electrical conductivity how are ionic bonds formed? Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other. Boiling and melting point 4.
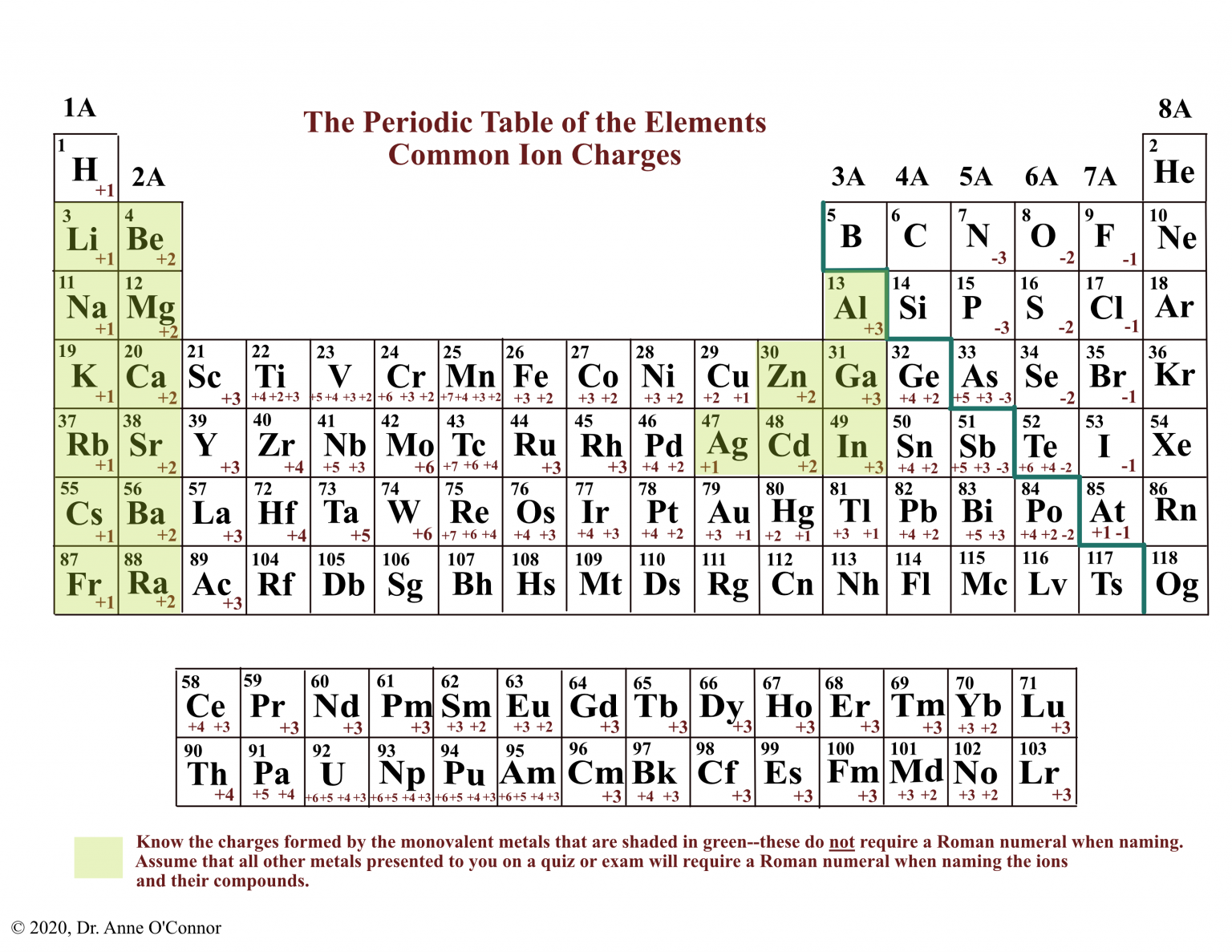
They form as a result of electrostatic attraction between oppositely charged ions and usually occur. Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other. Web the three ions would adhere (bond) to each other by the positive/negative attraction between the ions. The chemical bond that is formed between 2 2 atoms through the transfer of one or more electrons from the electropositive or metallic element. For example, sodium (na), a metal, and chloride (cl), a nonmetal, form an ionic bond to make nacl. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another. Predict what other elements might form ionic bonds. The alkali halides (nacl, lif, etc.). Web ionic bonding is the complete transfer of valence electron (s) between atoms. Web ionic bonds form when two or more ions come together and are held together by charge differences.
Boiling and melting point 4. Predict what other elements might form ionic bonds. These are electronegative elements with high ionization energies. Web ionic bonds are one of the two main types of chemical bonds. Web forming an ionic bond. Electrical conductivity how are ionic bonds formed? For example, sodium (na), a metal, and chloride (cl), a nonmetal, form an ionic bond to make nacl. They form as a result of electrostatic attraction between oppositely charged ions and usually occur. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another. An atom of sodium will lose an electron and form a positive ion.
Ionic Bond Definition, Types, Properties & Examples
Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other. Web ionic bonding is the complete transfer of valence electron (s) between atoms. Web metals and nonmetals are involved in ionic bonding, but it specifically refers to two elements that are oppositely charged being bonded to eachother..
2.6 Ionic Compounds and Formulas Chemistry LibreTexts
Web ionic bonds form when two or more ions come together and are held together by charge differences. This exchange results in a more stable, noble gas. For example, sodium (na), a metal, and chloride (cl), a nonmetal, form an ionic bond to make nacl. Web ionic bonds are one of the two main types of chemical bonds. So how.
Periodic Table Ions List Periodic Table Timeline
Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other. Predict what other elements might form ionic bonds. Boiling and melting point 4. So how do you know what kind of bond an atom will make? They form as a result of electrostatic attraction between oppositely charged.
Naming Simple Ionic Compounds Pathways to Chemistry
Web metals and nonmetals are involved in ionic bonding, but it specifically refers to two elements that are oppositely charged being bonded to eachother. Boiling and melting point 4. Web ionic bonding occurs in compounds composed of strongly electropositive elements (metals) and strongly electronegative elements (nonmetals). They form as a result of electrostatic attraction between oppositely charged ions and usually.
How Does An Ionic Bond Form Between Sodium And Chlorine slideshare
The chemical bond that is formed between 2 2 atoms through the transfer of one or more electrons from the electropositive or metallic element. It is a type of chemical bond that generates two oppositely charged ions. Web metals and nonmetals are involved in ionic bonding, but it specifically refers to two elements that are oppositely charged being bonded to.
Ionic Properties
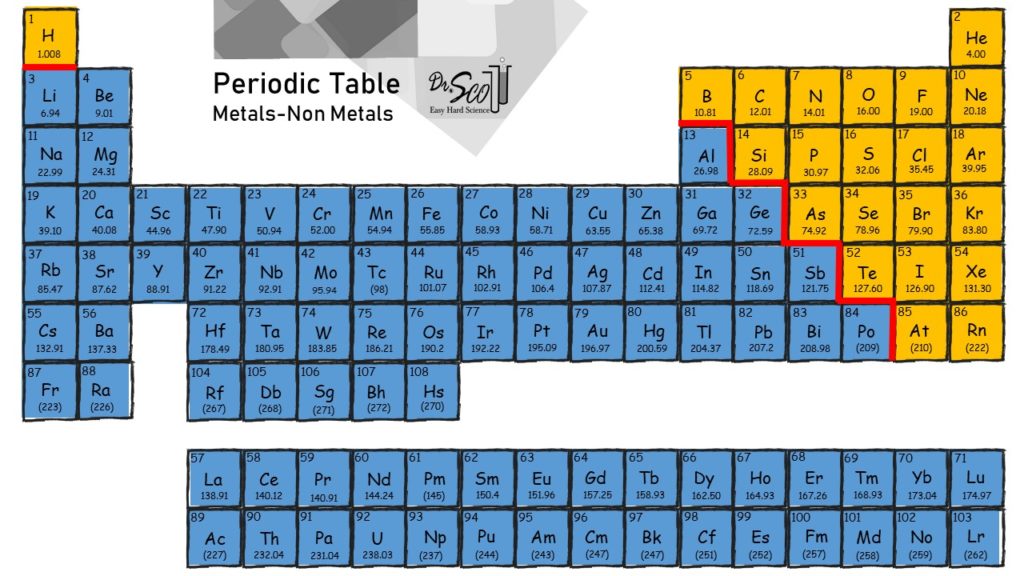
Web metals and nonmetals are involved in ionic bonding, but it specifically refers to two elements that are oppositely charged being bonded to eachother. Metals on the left and in the center of the periodic table form ionic bonds with nonmetals on the right of the. This exchange results in a more stable, noble gas. An atom of sodium will.
Ionic Bond Definition, Types, Properties & Examples
The chemical bond that is formed between 2 2 atoms through the transfer of one or more electrons from the electropositive or metallic element. Boiling and melting point 4. This exchange results in a more stable, noble gas. Web the three ions would adhere (bond) to each other by the positive/negative attraction between the ions. Web ionic bonding occurs in.
Ionic Bond Definition Easy Hard Science
Web ionic bonds are one of the two main types of chemical bonds. Web ionic bonding is the complete transfer of valence electron (s) between atoms. Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other. Ionic bonding is a type of chemical bond in which valence.
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
Electrical conductivity how are ionic bonds formed? Web forming an ionic bond. Boiling and melting point 4. Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other. Metals on the left and in the center of the periodic table form ionic bonds with nonmetals on the right.
Chemical Structure Chemical Bonding. Ionic, Metallic & Coordinate Bo…
Web ionic bonds form when two or more ions come together and are held together by charge differences. Boiling and melting point 4. For example, sodium (na), a metal, and chloride (cl), a nonmetal, form an ionic bond to make nacl. It is a type of chemical bond that generates two oppositely charged ions. Web metals and nonmetals are involved.
The Alkali Halides (Nacl, Lif, Etc.).
So how do you know what kind of bond an atom will make? Web the three ions would adhere (bond) to each other by the positive/negative attraction between the ions. Boiling and melting point 4. They form as a result of electrostatic attraction between oppositely charged ions and usually occur.
Electrical Conductivity How Are Ionic Bonds Formed?
Web forming an ionic bond. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another. Predict what other elements might form ionic bonds. Web ionic bonds are one of the two main types of chemical bonds.
It Is A Type Of Chemical Bond That Generates Two Oppositely Charged Ions.
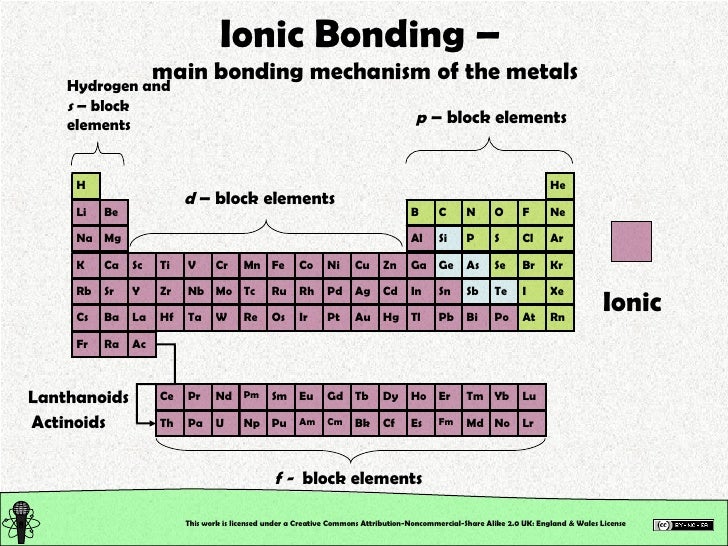
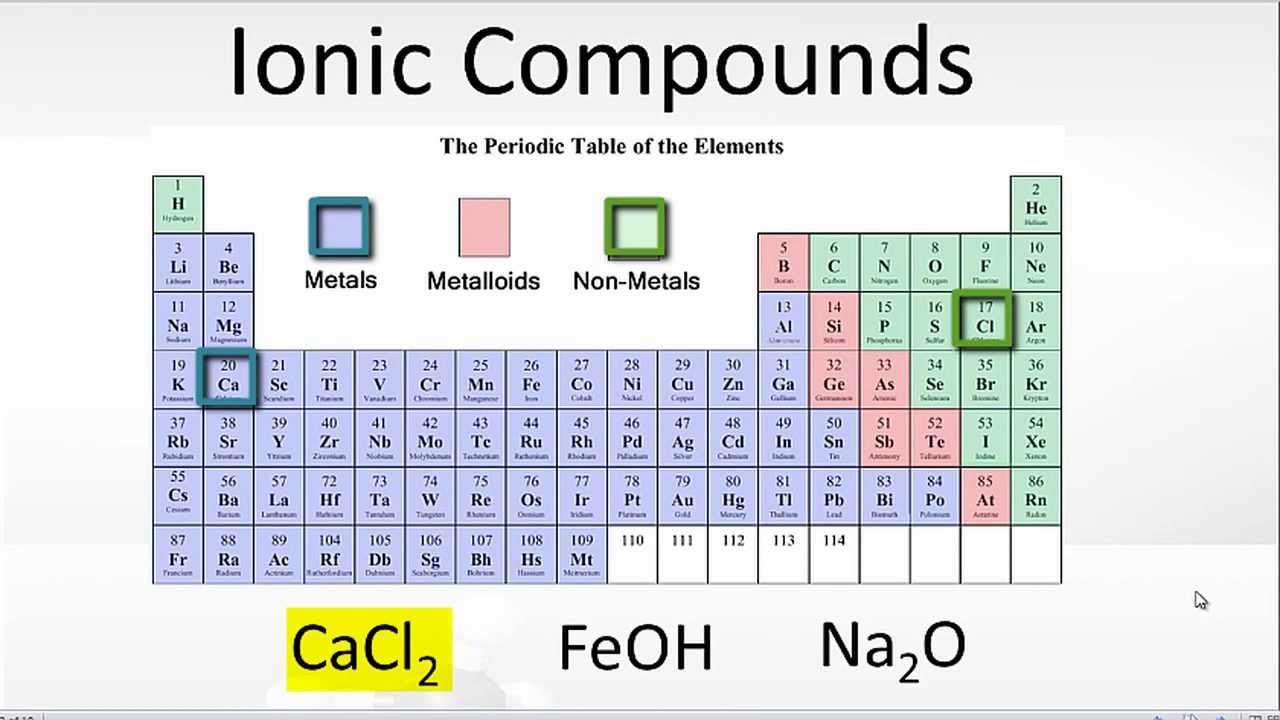
The chemical bond that is formed between 2 2 atoms through the transfer of one or more electrons from the electropositive or metallic element. Web ionic bonding occurs in compounds composed of strongly electropositive elements (metals) and strongly electronegative elements (nonmetals). Web metals and nonmetals are involved in ionic bonding, but it specifically refers to two elements that are oppositely charged being bonded to eachother. Metals on the left and in the center of the periodic table form ionic bonds with nonmetals on the right of the.
Web Ionic Bonds Usually Occur Between Metal And Nonmetal Ions.
This exchange results in a more stable, noble gas. An atom of sodium will lose an electron and form a positive ion. Web ionic bonds form when two or more ions come together and are held together by charge differences. Web stephen lower simon fraser university learning objectives explain the fundamental difference between the bonding in metallic solids compared to that in other.

.PNG)